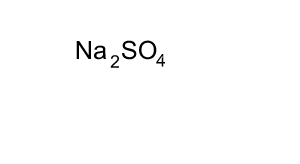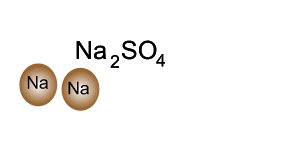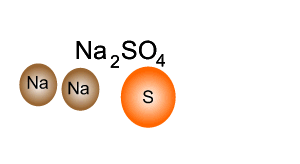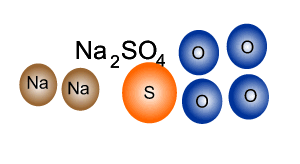Q12 Write a balanced molecular equations for the word equations 1 Calcium Oxygen Calcium oxide 2 Cal...

Chalk contains 10 % Calcium, 3 % Carbon and 12% Oxygen. Find the amount (in grams) of each of these compounds in 1 kg of chalk.

Composition of the human body: Oxygen 65%, Carbon 18%, Hydrogen 10%, Nitrogen 3%, Calcium 1.5%, Phosphorus 1%, Potassium 0.25%, Sulphur 0.25%, Sodium 0.15%, Chlorine 0.15% Magnesium 0.05%, Iron 0.006%, Fluorine 0.0037%, Zinc

Classification of Matter Classification of Matter Matter 1. Pure Substance a. Elementb. Compound 2. Mixtures a. Homogeneous b. Heterogeneous Combine to. - ppt download

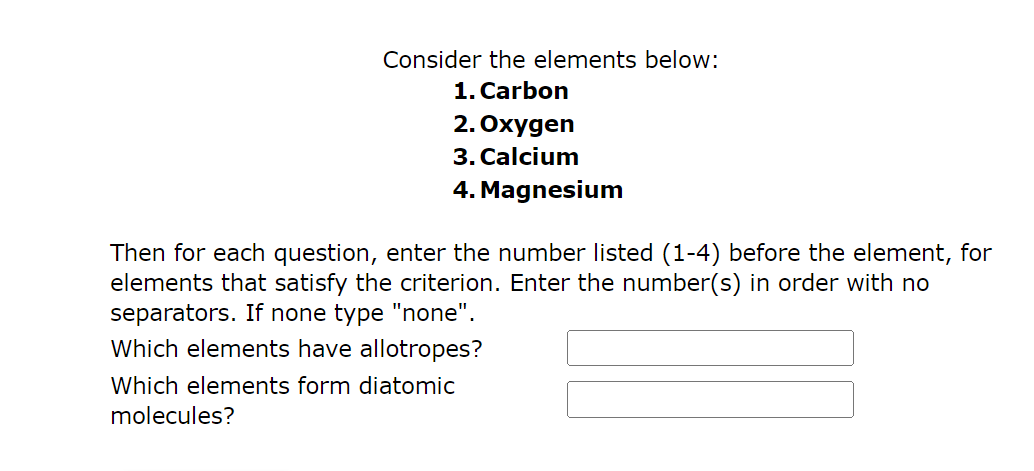
Activity 3. Let's Identify Objective: Identify the atoms present in the common compounds Directions: - Brainly.ph
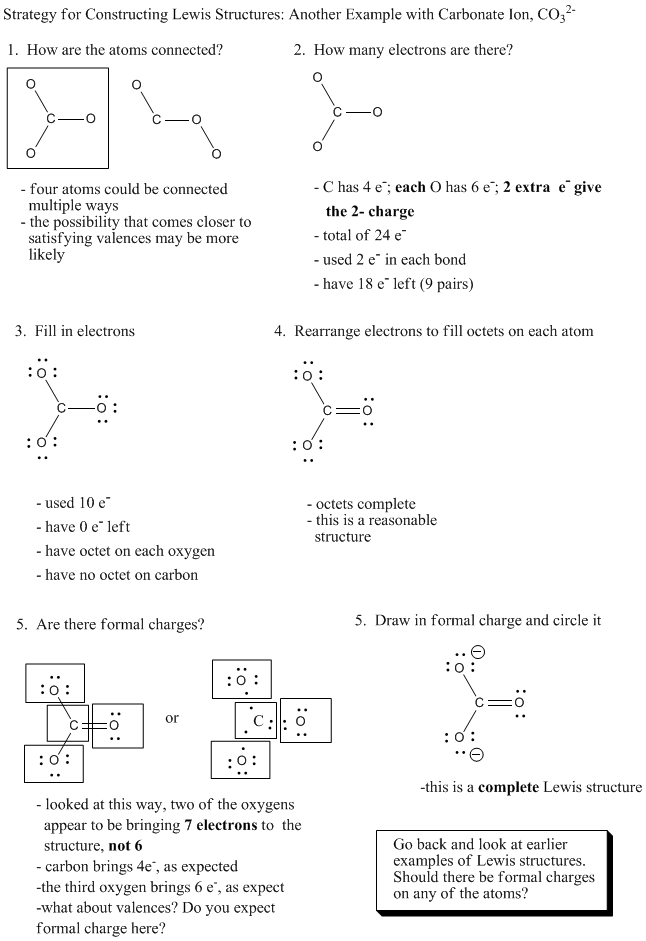
Which of the following has a greater carbon oxygen bond length? 1. Sodium formate 2. Calcium carbonate

Question Video: Calculating the Mass of Calcium Carbonate Required to Produce a Given Mass of Calcium Oxide | Nagwa

Chalk contains 3% of carbon, 10% of calcium and 12% of oxygen. Find the amount in grams of each of these substance in 1 kg of chalk.

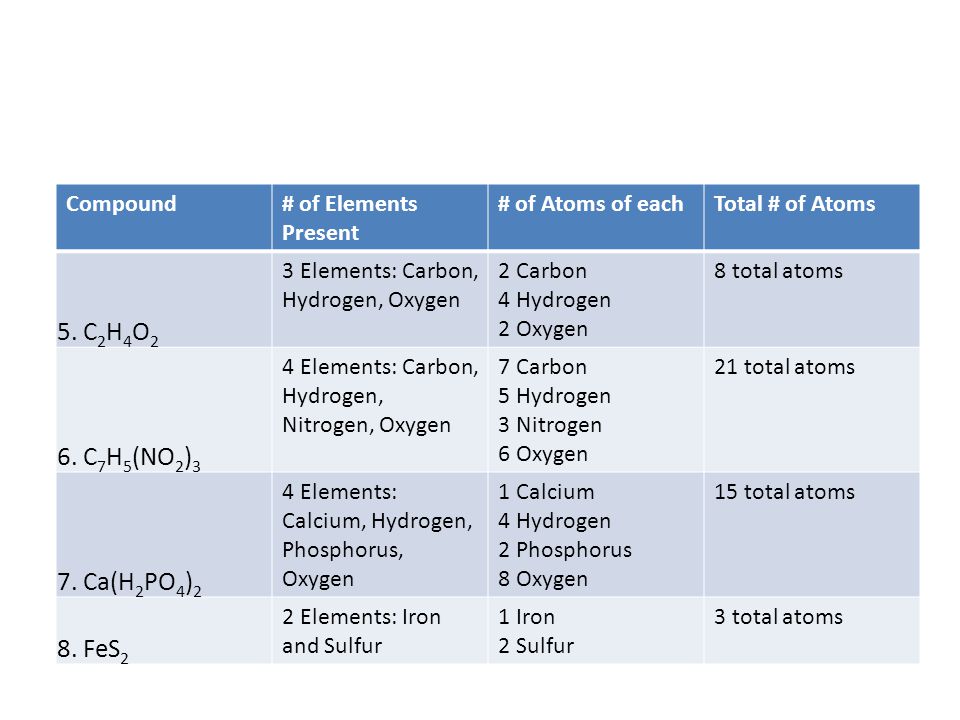
Page 30: Counting Elements and Atoms Practice Page 31: Review of Elements and Compounds. - ppt download