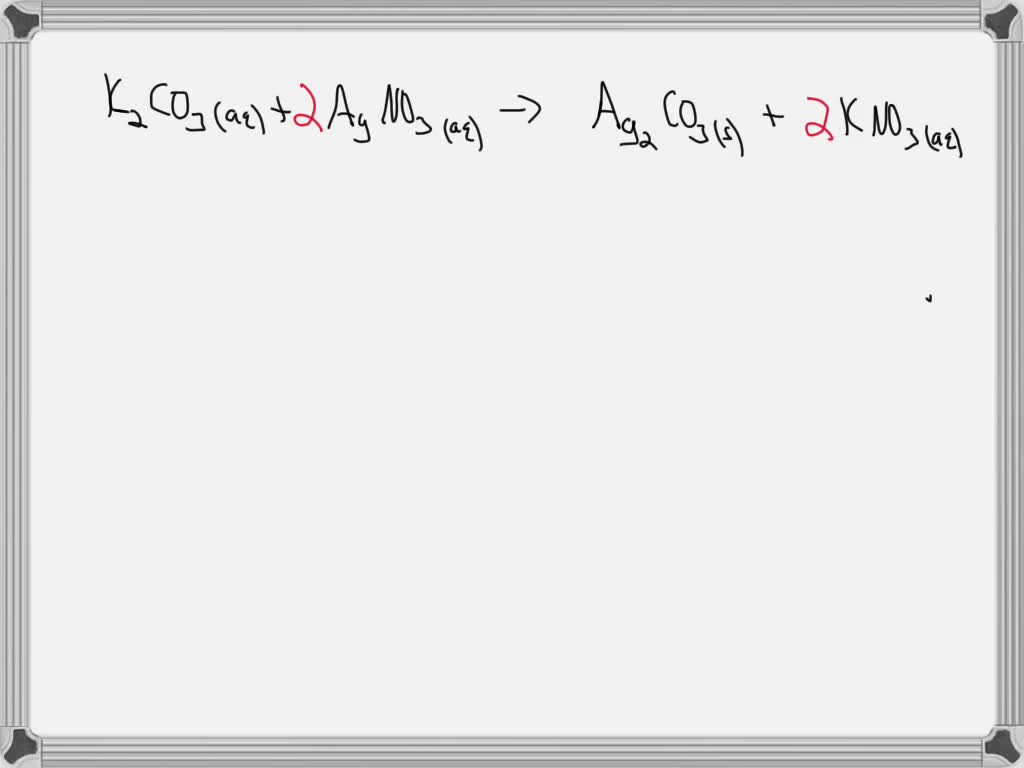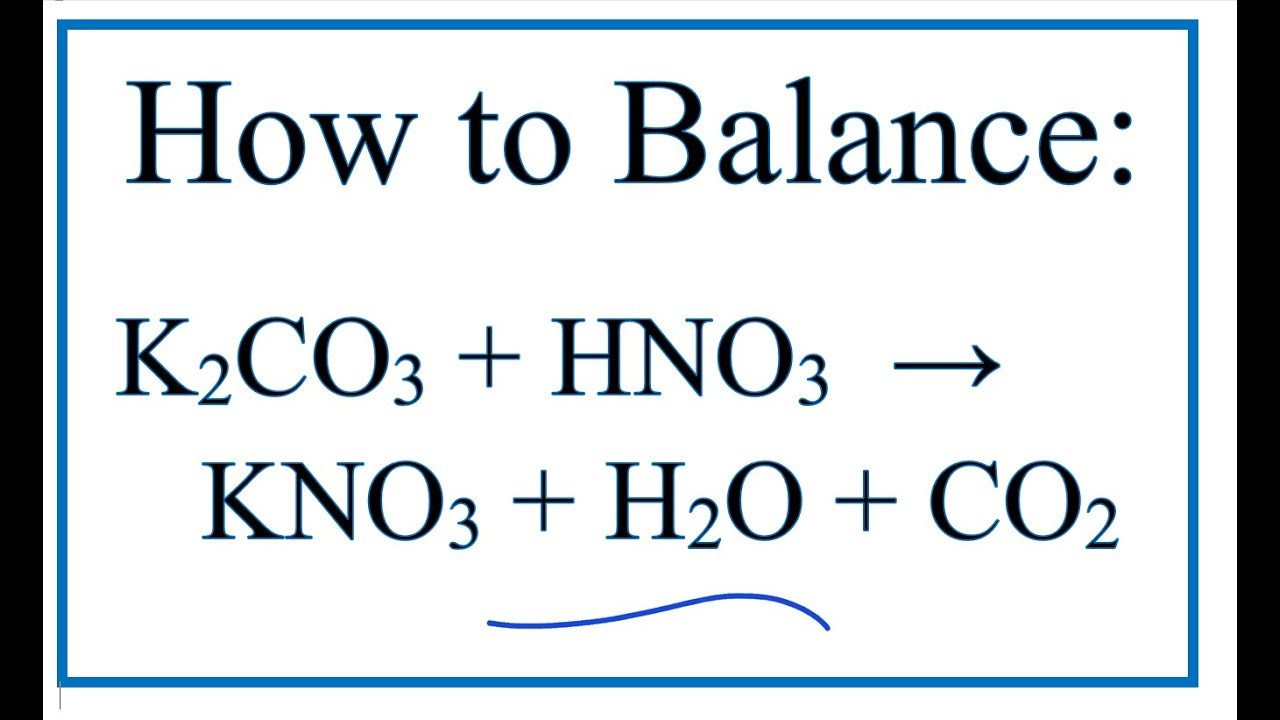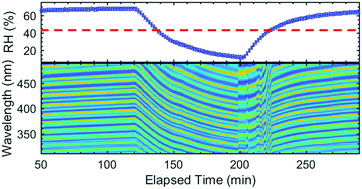
Phase transition dynamics of single optically trapped aqueous potassium carbonate particles - Physical Chemistry Chemical Physics (RSC Publishing)

SOLVED:Aqueous barium chloride and aqueous potassium carbonate react to produce solid barium carbonate and aqueous potassium chloride.

Density and Solubility of CO2 in Aqueous Solutions of (Potassium Carbonate + Sarcosine) and (Potassium Carbonate + Pipecolic Acid) | Journal of Chemical & Engineering Data
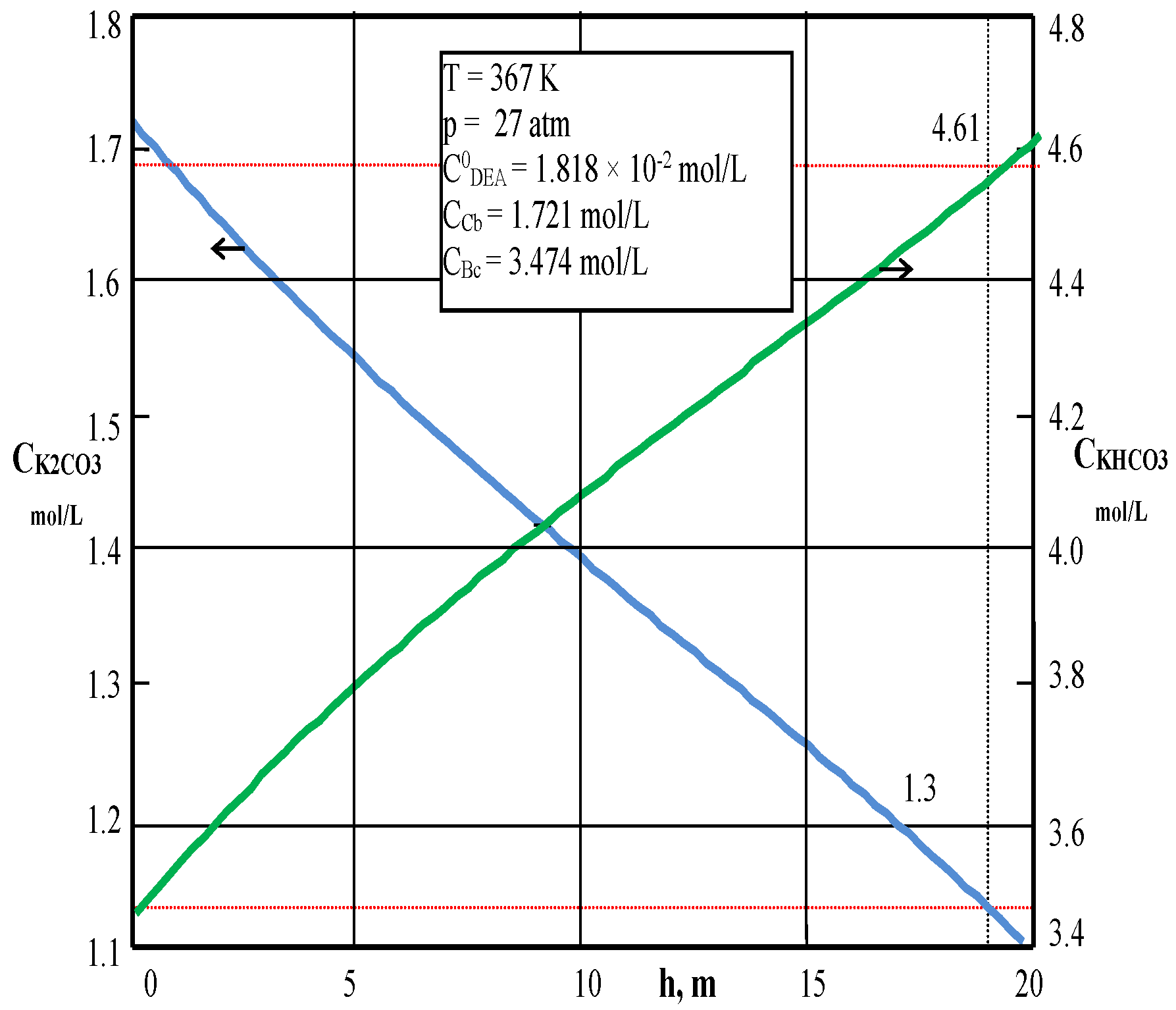
Processes | Free Full-Text | New Approaches in Modeling and Simulation of CO2 Absorption Reactor by Activated Potassium Carbonate Solution

Kinetics of CO2 Absorption Into Aqueous Potassium Carbonate Solution Promoted by Carbonic Anhydrase Enzyme | AIChE
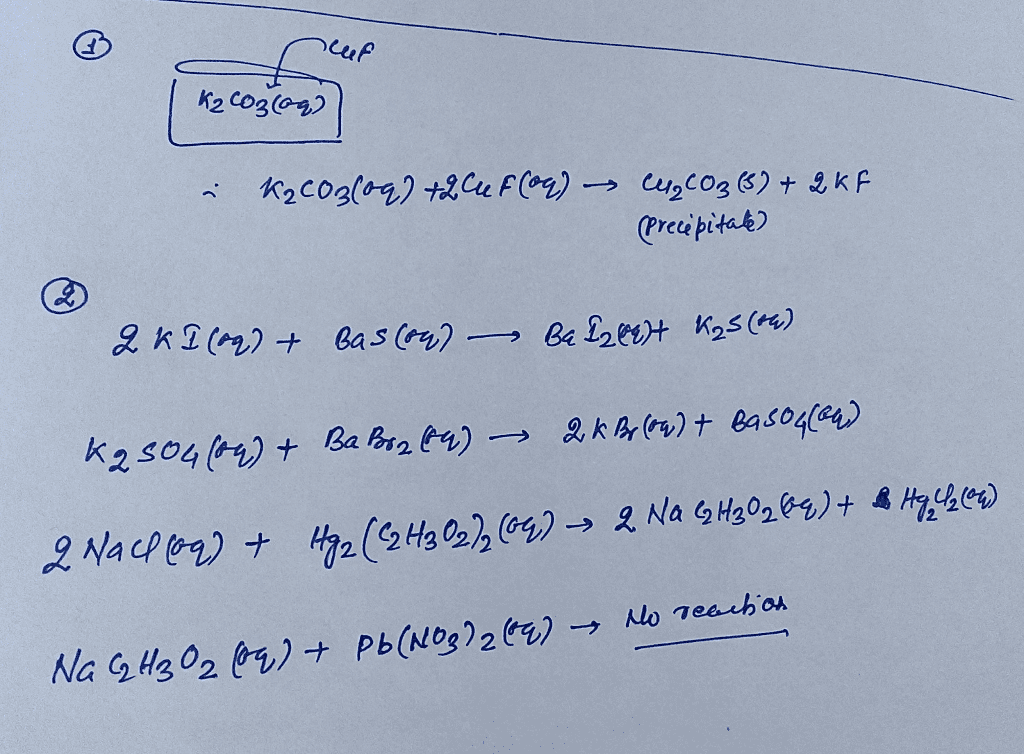
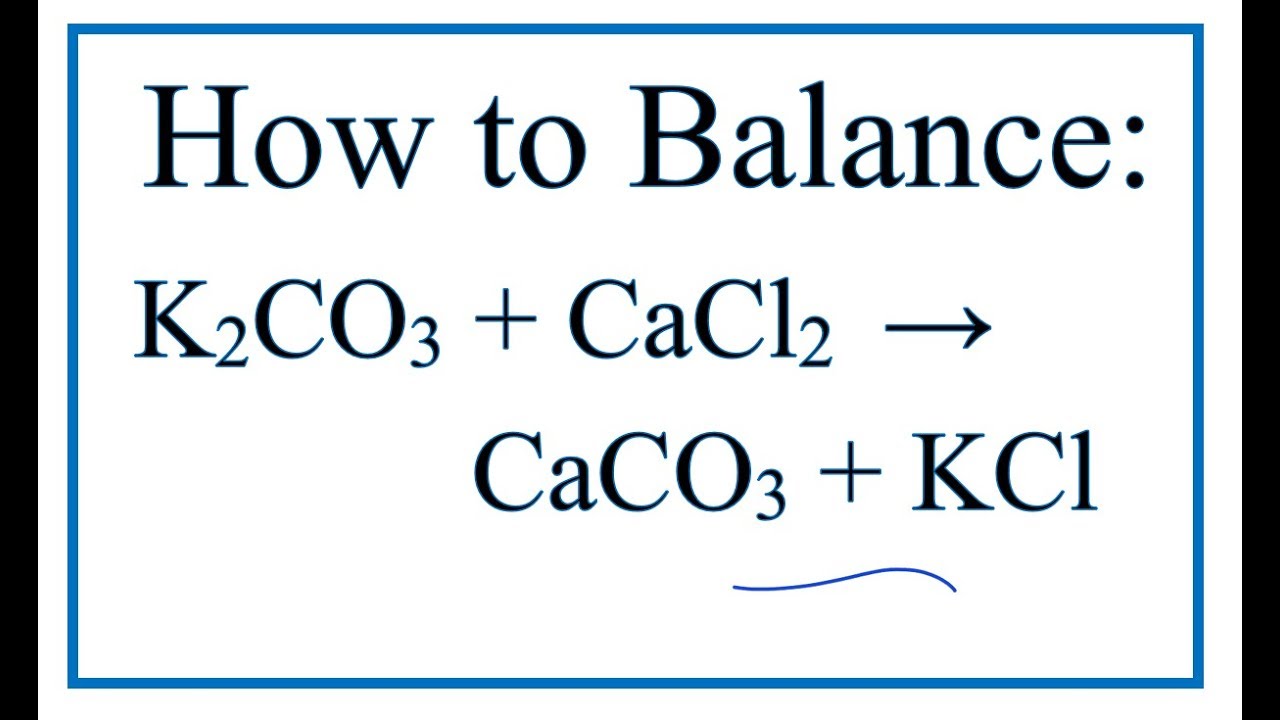
OneClass: 1. Aqeous potassium carbonate was mixed with aqueous copper (I) fluoride, and a crystallize...

Catalytic Hydroboration of Aldehydes, Ketones, and Alkenes Using Potassium Carbonate: A Small Key to Big Transformation | ACS Omega

Preparation of a potassium carbonate (K2CO3) solution for microplastic... | Download Scientific Diagram

pH-values of potassium carbonate solutions with different amounts of... | Download Scientific Diagram