
Write a balanced chemical equation for the reaction of calcium carbonate and dil. hydrochloric acid.

SOLVED: Write net ionic equation for the reaction that occurs when excess hydrochloric acid (aq) and barium carbonate are combined Write net ionic equation for the reaction that occurs when excess hydroiodic

Question Video: Using Strong Acid-Strong Base Titration Data to Calculate the Concentration of the Base | Nagwa
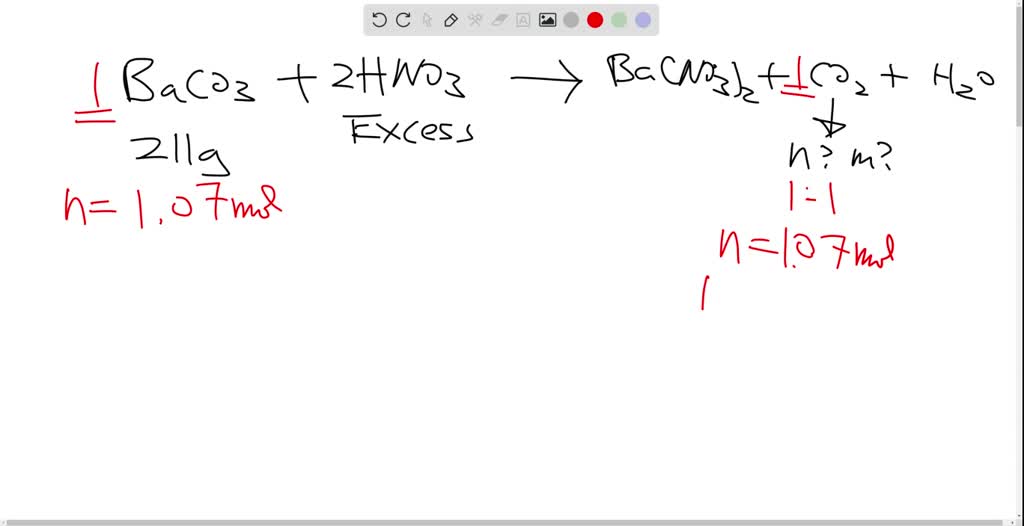
SOLVED:A 211 g sample of barium carbonate, BaCO 3, reacts with a solution of nitric acid to give barium nitrate, carbon dioxide and water. If the acid is present in excess, what
What is the balanced net-ionic equation for the gas producing reaction between hydrobromic acid and solid calcium carbonate? - Quora

SOLVED: Experimental Section 1,Carbonate Ion 1. Pour of" sodium carbonate solution t0 lest tubeand add several drops of barium chloride. 1.2 Afier sedimentalion remove the solution over resulting precipitate and add several

Titration of the sodium hydroxide solution with adsorbed CO 2 after... | Download Scientific Diagram











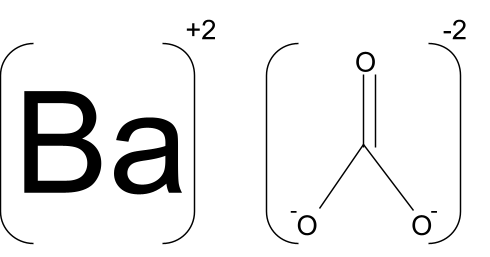



![Barium Carbonate [BaCO3] Molecular Weight Calculation - Laboratory Notes Barium Carbonate [BaCO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/barium-carbonate-molecular-weight-calculation-300x205.jpg)