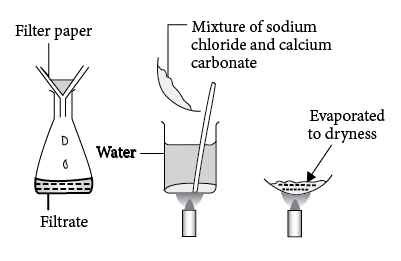
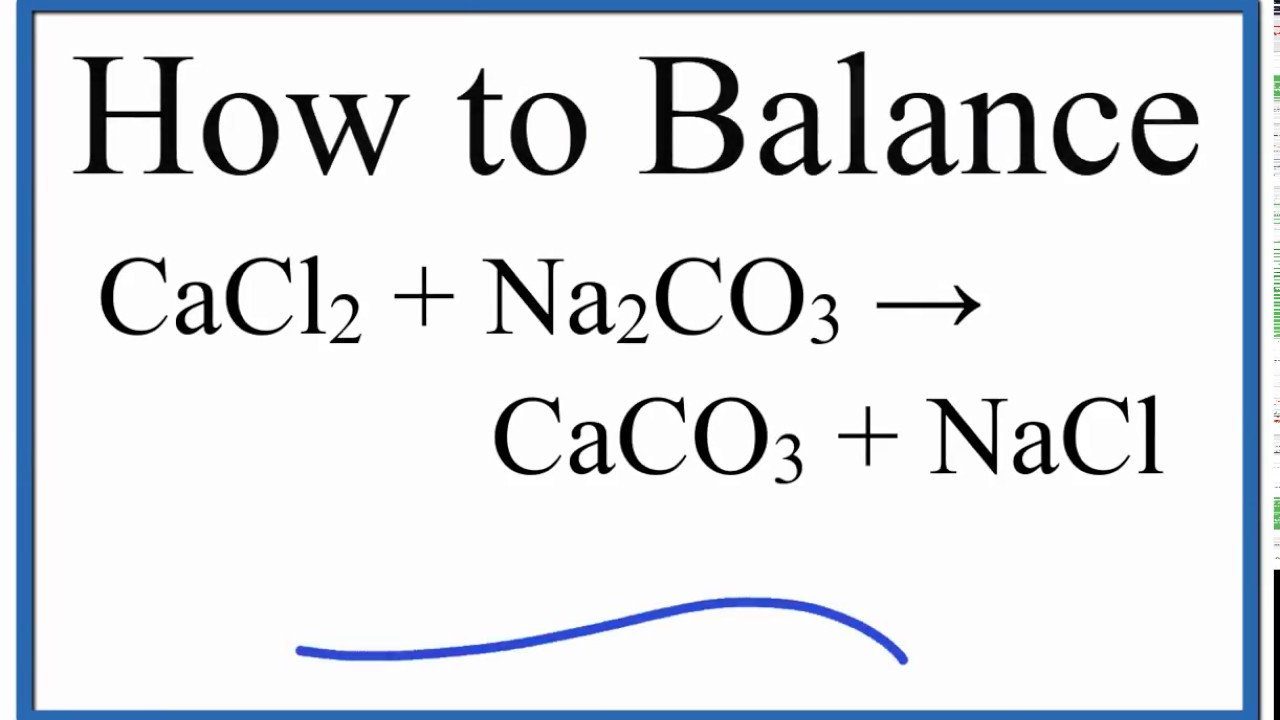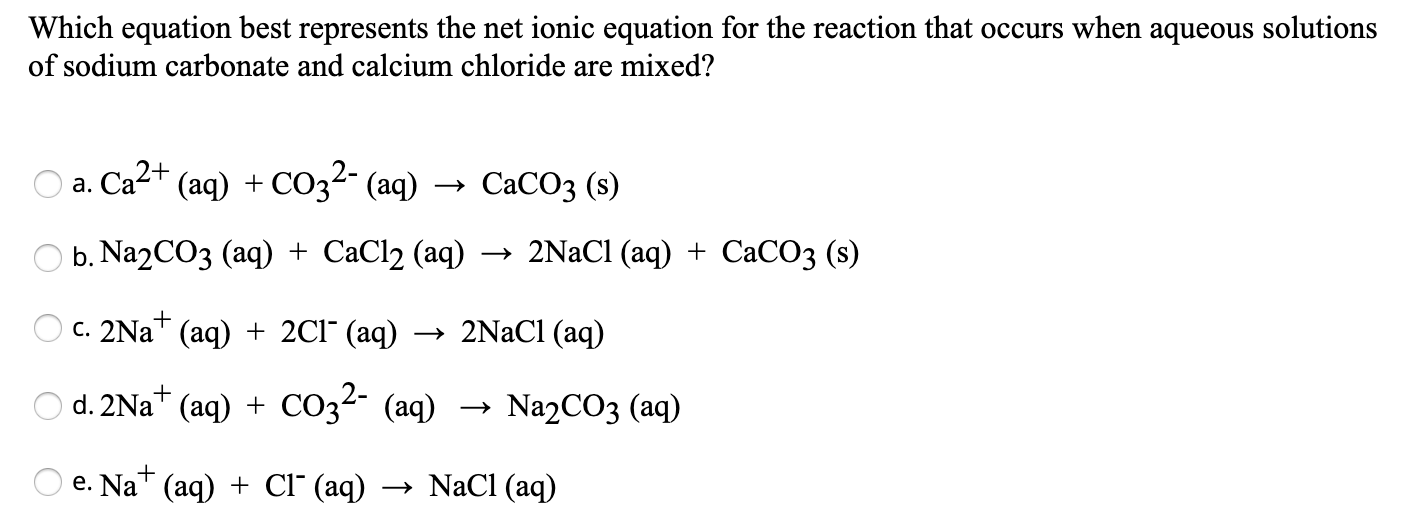Composition inversion to form calcium carbonate mixtures - CrystEngComm (RSC Publishing) DOI:10.1039/C7CE00433H
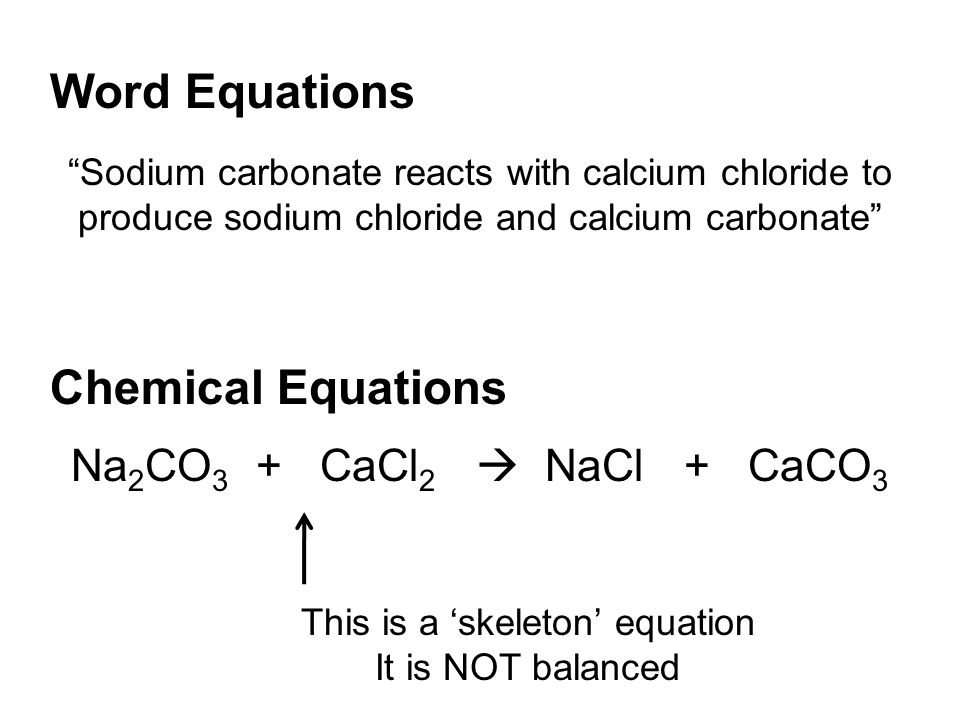
Chapter 11 Chemical Reactions Anything in black letters = write it in your notes ('knowts') UNIT 4 – Chemical Names, Formulas & Rxns Chapter 9 – Chemical. - ppt download

What precipitate will form when aqueous solutions of sodium carbonate calcium Na_2CO_3 and calcium chloride CaCl_2 are mixed? | Socratic
111 gram sample of a mixture of calcium chloride and Sodium Chloride is treated with sodium carbonate to precipitate precipitate as Calcium precipitate this kkcalcium carbonate on heating gives 14 gram of

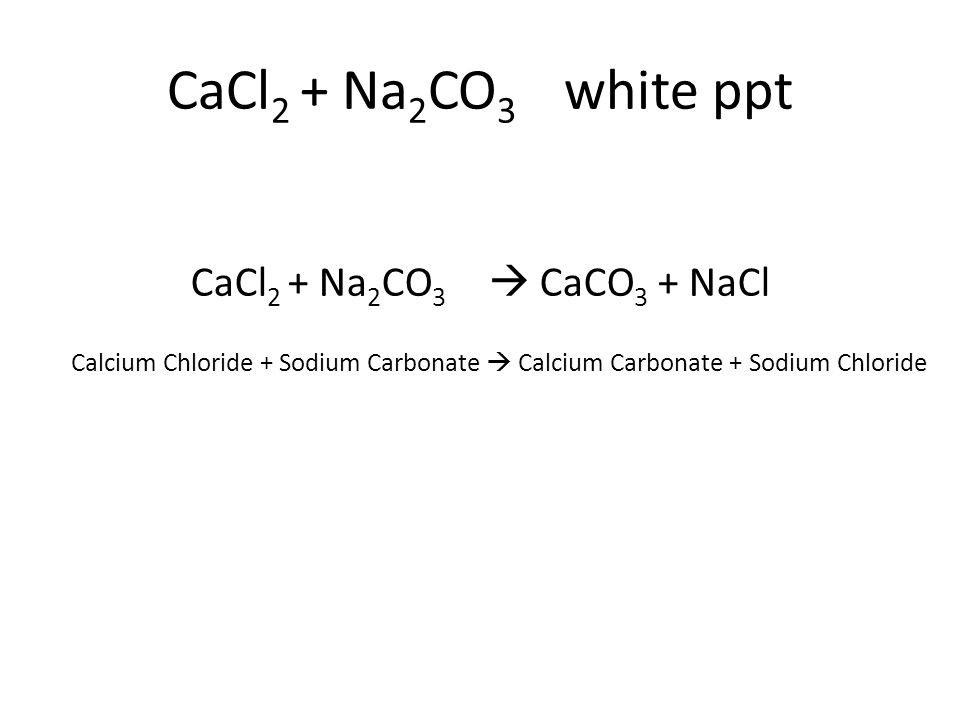
What happens when CaCl2 reacts with Na2CO3 | CaCl2 + Na2CO3 | Calcium chloride + Sodium carbonate - YouTube

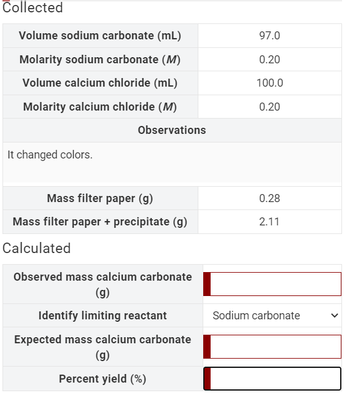
SOLVED: CaCl(aq) Na.CO (aq) CaCO (s) 2NaCl(aq) Could ammonium carbonate solution have been used in lieu of sodium carbonate solution? If so, assuming the concentrations of 0.110 M sodium carbonate and 0.100

SOLVED: 4.Consider a reaction between sodium carbonate (MW: 105.99 glmol) ad calcium chloride (MW: 110.98 g/mol): The products formed are calcium carbonate and: sodium chloride CaCh Na COs CaCO,+ ZNaCl How many