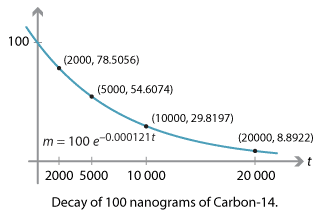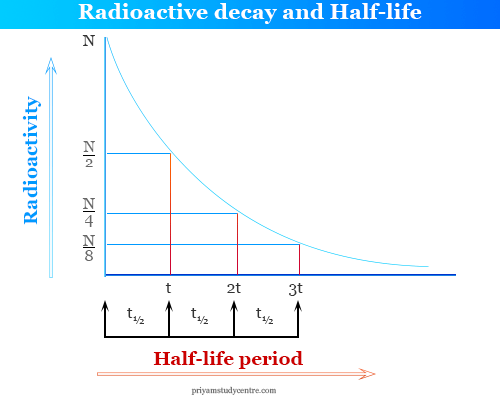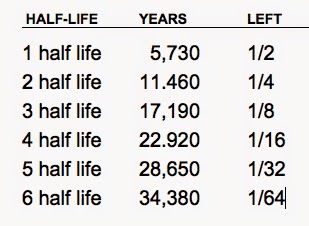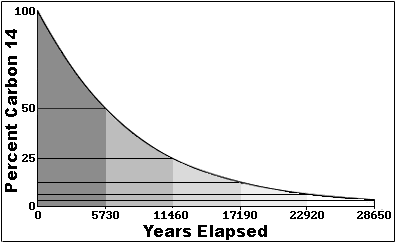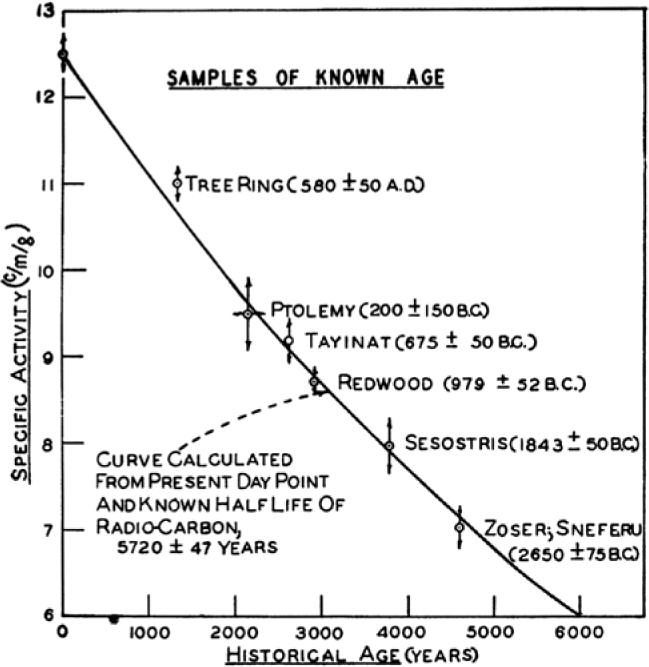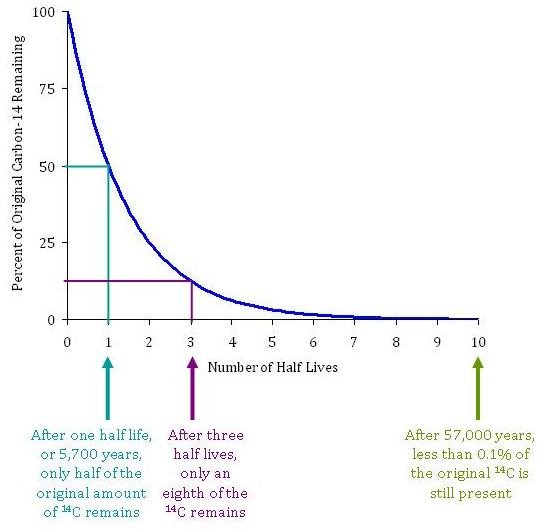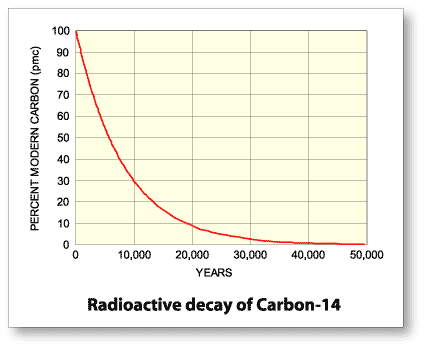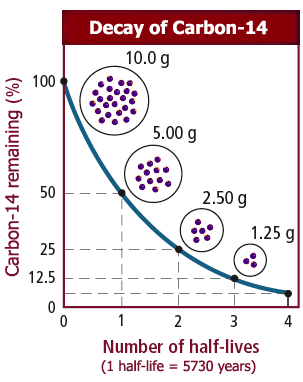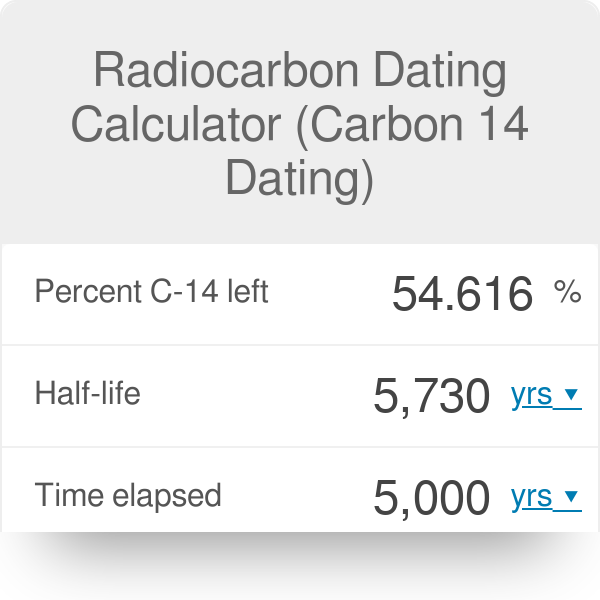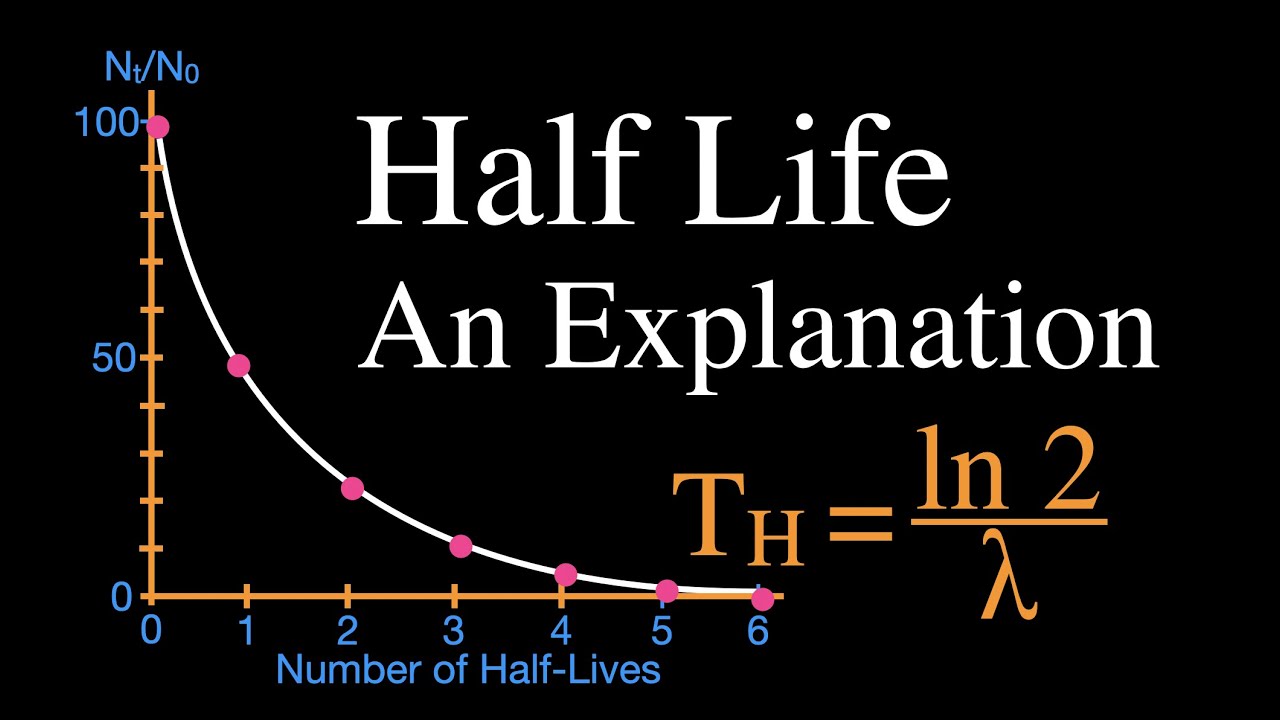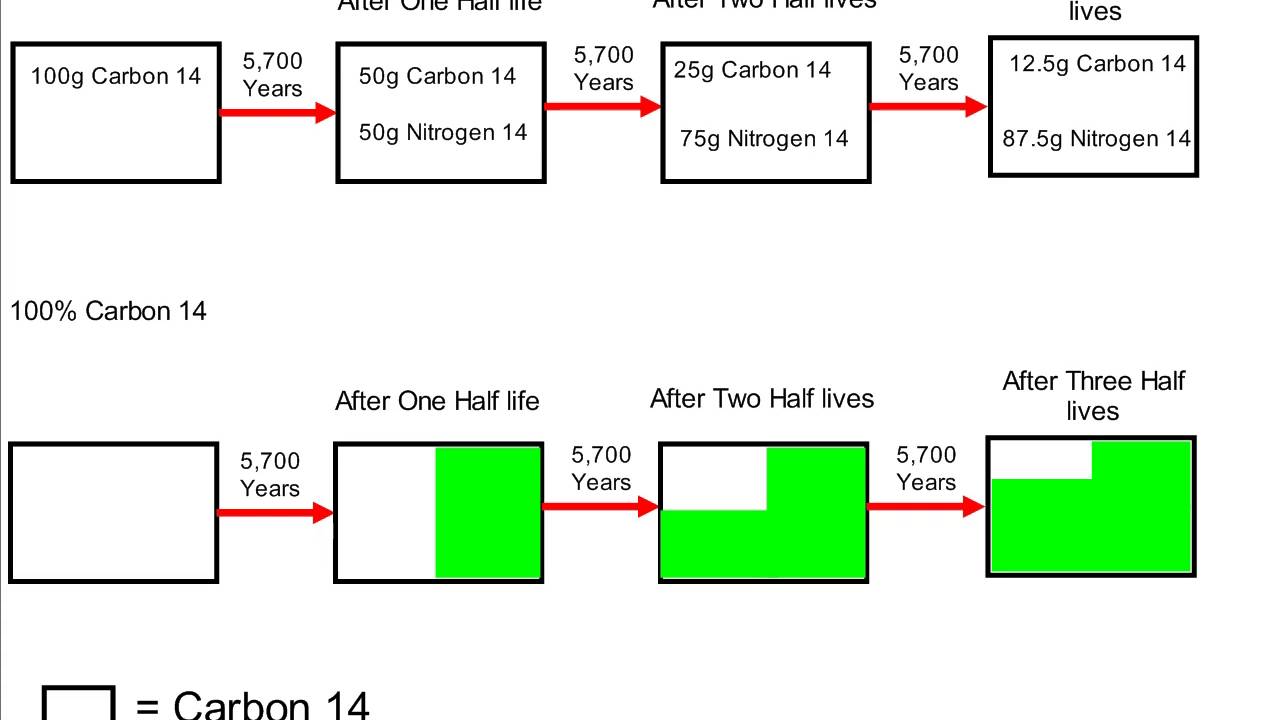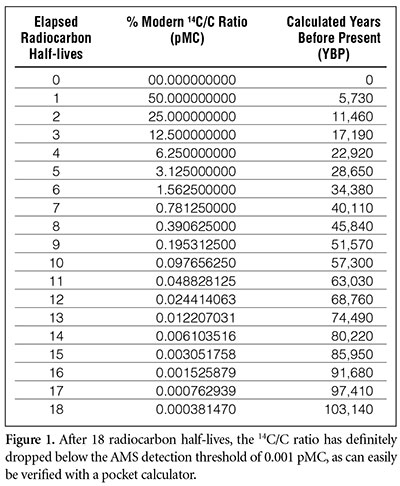
Rethinking Carbon-14 Dating: What Does It Really Tell Us about the Age of the Earth? | The Institute for Creation Research
The half-life of carbon-14 is 5,730 years. If the original sample is 200 grams, how much of a given amount remains after 22,920 years? - Quora
How do we know that the half life of carbon 14 is 5730 years? And how does finding out the ration between c12 and c14 pertain to radiocarbon dating? - Quora

EduMission: Physics Form 5: Chapter 5 - Application of Radioactive (Carbon-14 Dating) | Dating, Assumptions, Physics
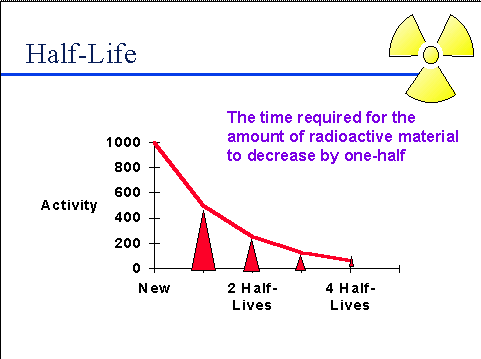
If carbon 14 has a half life of 5,730 years and a sample contains 70 mg originally, how much is left after 17,190 years? | Socratic
If Earth is billions of years old, why is there still carbon-14 in rocks when C14 takes only 5000-ish years to totally decay? - Quora