Single Displacement Reaction - Iron Nail in Copper Sulfate Solution. Types of Chemical Reactions, Part 2 of 7 Stock Vector - Illustration of liquid, background: 94216261
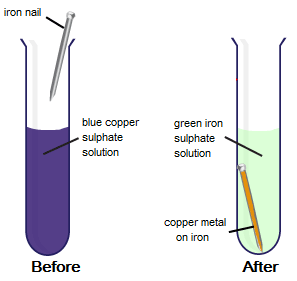
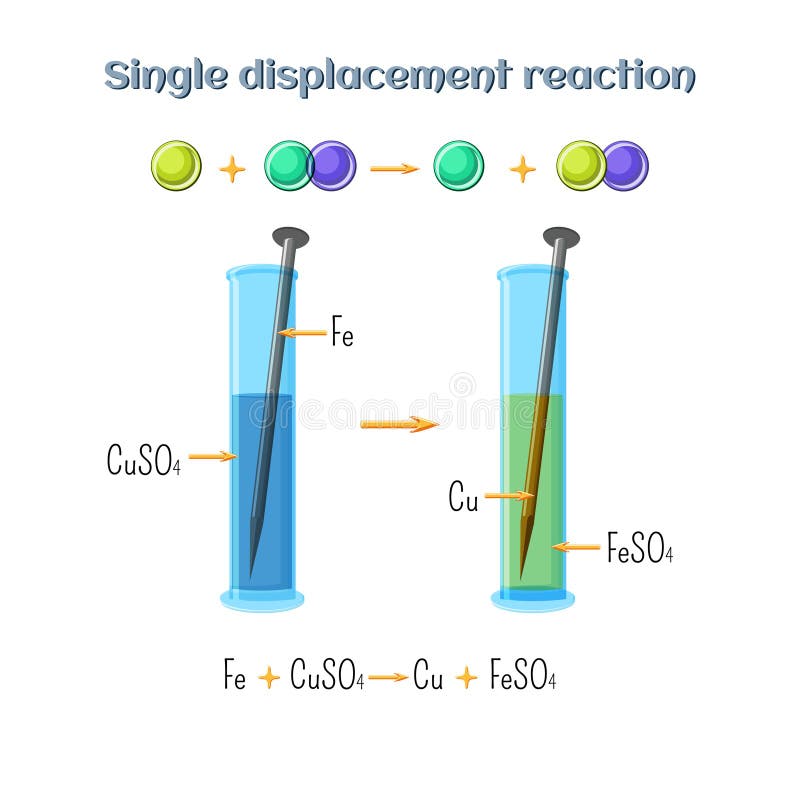
Why does the colour of copper sulphate solution change when an iron nail is dipped in it. - Sarthaks eConnect | Largest Online Education Community

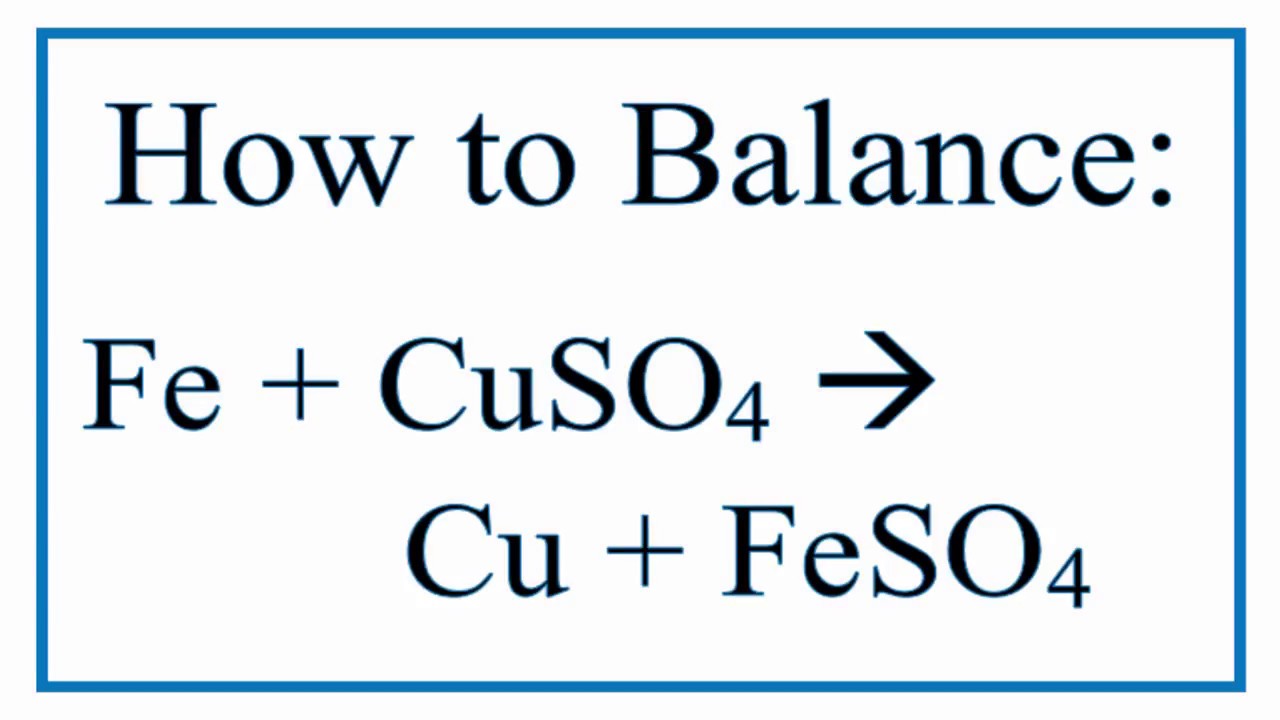
Purpose – To react copper sulfate with iron, producing iron sulfate and copper. Materials – copper sulfate (CuSO 4 ) and Fe filings – water – beakers, - ppt download
Joanne added iron filings to copper sulphate solution. She observed the reaction after one week. (a) What evidence in the di

Class 9 Chemistry Practical's and Experiments on To carry out the reaction of Iron with copper sulphate solution in water and classify it as physical or chemical changes

What are Ores? Most metals are too …………… to exist on their own in the ground. Instead they exist combined with other elements (typically o……….. or sulphur) - ppt download