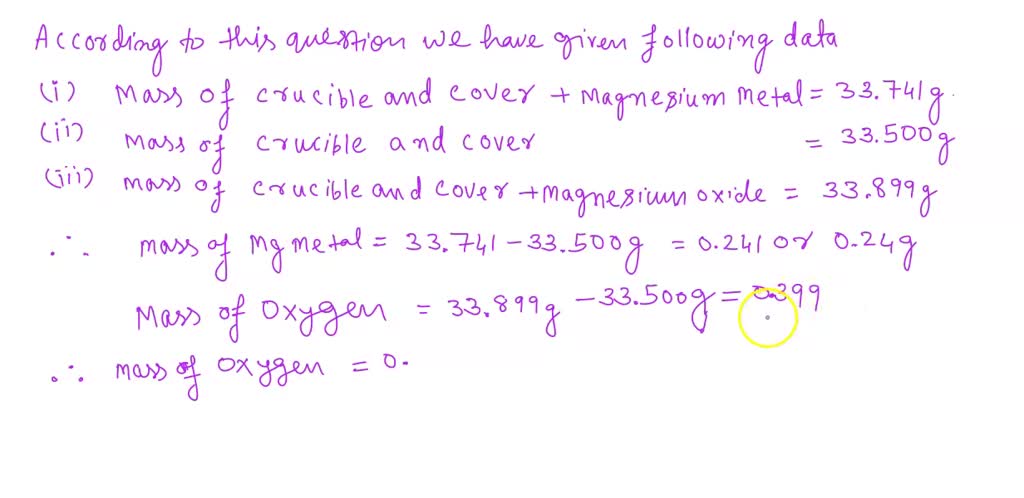SOLVED: Table 1. The Reaction of Magnesium Ribbon Measurement Trial Mass cruciblelg 31.06 Mass of Mg ribbonlg Mass of crucible and Mg ribbonlg) 31.63 Mass crucible and procuct (after heating) (g 31.97

Magnesium Ribbon Stock Illustrations – 6 Magnesium Ribbon Stock Illustrations, Vectors & Clipart - Dreamstime

1.Define displacement reaction. 2.Predict what will happen when: a)Magnesium is put into Silver Nitrate solution b)Zinc is put into Magnesium Nitrate solution. - ppt download
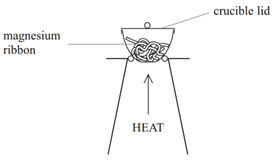
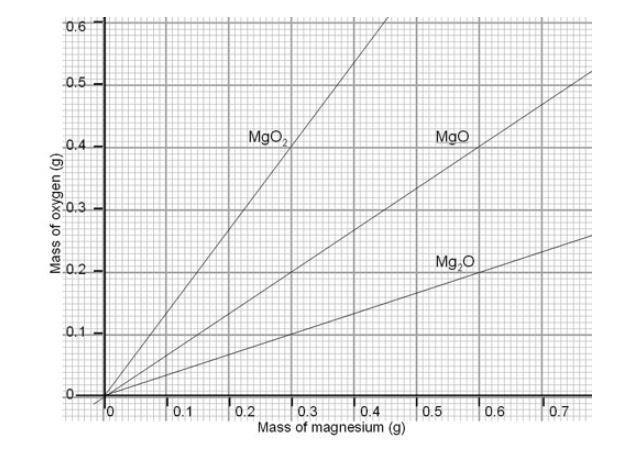
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry

Magnesium Ribbon Rolls- 99.95% Pure Magnesium Ribbon for Science and Lab Experiments, by American Heritage Industries : Amazon.ca: Health & Personal Care

9.3 Magnesium ribbon burns in air.A. Write the chemical symbol for magnesium.B. Name the element in - Brainly.in

Write the balanced chemical equations for the following reactions.(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water(b) Zinc + Silver nitrate → Zinc nitrate + Silver(c) Aluminium + Copper chloride

How to write a Chemical Reaction in Chemical Equation Form? | Chemical equation, Chemical reactions, Chemical