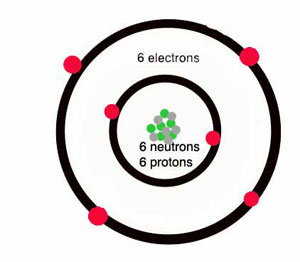
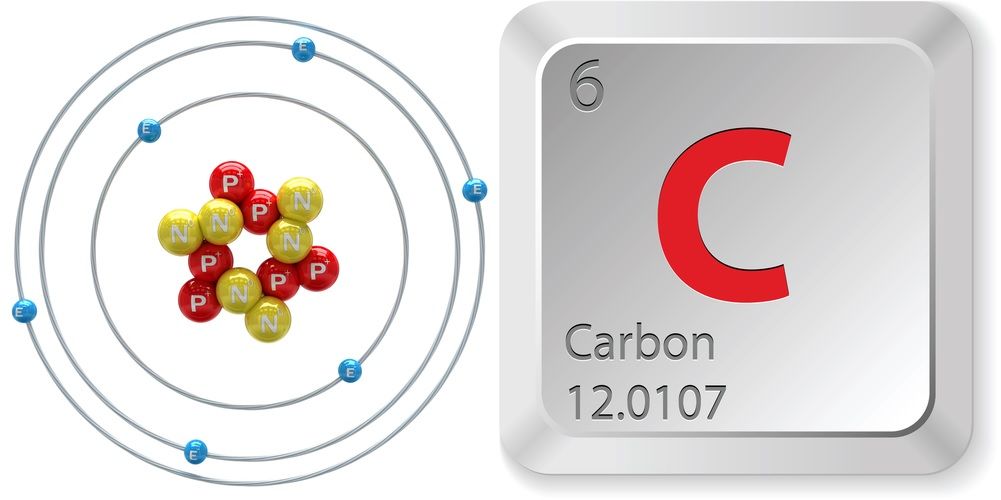
1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

If one mole of carbon atoms weighs 12 grams, what is the mass (in grams) of 1 atom of carbon?... - YouTube

a) How many atoms are there in exactly 12g of carbon-12 element ? `(C = 12 u)` (b) What name is - YouTube
16. How many atoms of carbon are present in a diamond weighing 0.5 carot ? (One carot weighs 200mg and assume the diamond to be pure carbon )

Calculate the difference in the number of carbon atoms in 1 0 g of C-14 isotope and 1 0 g - Chemistry - Some Basic Concepts of Chemistry - 13730077 | Meritnation.com

Calculate the number of atoms in (i) 0.5 mole atoms of carbon (C^12) (ii) 3.2 g of sulphur (iii) 18.0 g of glucose (C6 H12O6) (iv) 0.20 mole molecules of oxygen

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5